Notation: Orbital and Lewis Dot
Since we're experts at figuring out electron configuration, let's explore another notation called orbital notation. In a sense, the orbital notation is very similar to the ways we learned to represent electron configurations in the previous section. Truth be told, it's just an alternative method to draw the configuration in a way to easily identify how the element can bond to other elements.
The orbital notation also helps us figure out what's really going on inside all of those crazy orbitals. For instance, when we see the electron configuration "3p5," we know that there are 5 electrons in the p subshell, but we don't know which orbitals the electrons are actually located in. Not to worry, the orbital notation provides us with that level of detail that simply can't be found in the electron configuration.
Let's get started. First we need to do the electron configuration of an atom. Want to use silicon as an example? We hope so.
1s22s22p63s23p2
First let's recall:
• s subshells have 1 orbital
• p subshells have 3 orbitals
• d subshells have 5 orbitals
• f subshells have 7 orbitals
For each subshell we'll first draw the corresponding number of orbitals (circles) above them as shown here:

Next we'll fill in the electrons represented as arrows. Don't forget Hund's Rule when filling in your electrons. Remember that the most stable arrangement for electrons in each subshell is the one with the greatest number of parallel spins, so we will distribute electrons evenly to orbitals with a given subshell first before any pairing occurs. In other words, each circle gets one arrow before any circle gets two arrows.

There are two other ways that we might see the orbital notation represented:
1) Where the orbitals are represented as rectangles and the electrons are represented as arrows.

2) Where the orbitals are represented as circles and the electrons are represented as slashes.

Now presenting, direct from Berkley, California: Lewis and his Amazing Dots. Lewis dot notation was first presented by Gilbert Newton Lewis in 1916 in an article entitled "The Atom and the Molecule" published in the Journal of the American Chemical Society.

This is going to look simple. That's because it is simple; thanks to good ol' Gilbert. Lewis was the first person we know to have done this and make it so obviously simple for the rest of us. Good job, Lewis.
This notation is just another way of showing details about an atom's valence (outer) shell. Unlike the electron configuration, which includes all of the electrons an atom has to offer, Lewis dots are only concerned with electrons in the outer most shell of an atom. This means that no matter how many electrons an element contains, Lewis dots will never number higher than eight. If it helps, just remember that super hip TV show from 1977 Eight is Enough.
Let's go back to silicon as an example. We've already learned the electron configuration of silicon is 1s22s22p63s23p2. The first (n = 1) and second (n = 2) shells are completely filled. The third (n = 3) shell has a filled s orbital but it's p and d orbitals remain empty. Therefore the third shell is our valence shell and the electrons in this shell are the valence electrons.
The Lewis dot notation consists of the kernel (the nucleus and all of its inner electrons that is represented by the elemental symbol) and the valence electrons represented by dots. Check out the lewis dot structure of carbon below:

The letter "C" is the kernel, and as we said this represents the atom and its inner shell electrons (the 1s2 electrons). The remaining four valence electrons are placed around the kernel according to specific rules. Don't worry about the rules just yet. We'll get to them soon. Each of the four sides around the elemental symbol represents one of the orbitals in the outer shell. Here are the rules:
Rule #1. No side can have more than two dots because each orbital can only hold two electrons.
Rule #2. When filling the sides of the element symbol each side gets one dot before doubling up…that's Hund's Rule, not ours. The exceptions to this rule are hydrogen and helium since their outer shells can only hold two electrons. Every other element obeys the second rule.
How many dots should we place around each element? The answer is surprisingly simple. Just take a look at the periodic table below. Remember how each dot is supposed to represent the number of electrons in the elements outer shell? Well, the periodic table displays the elements in a way for us to easily determine how many electrons are in their outer shell.
The column in which the element is located will tell us how many valence electrons each element has in its outer shell. Remember hydrogen and helium can only have a maximum of 2 valence electrons in their outer shell so they are the exceptions to the rule.

Let's see some examples:

As we go straight across the second row of the periodic table we keep adding one more dot or electron (clockwise) to each element. We can do this all the way to Ne, the noble gas, which has a full octet and is quite pleased about it, too.
Keep in mind it doesn't matter where we first put our dot around the element. Just make sure you choose a side and then start adding your dots in either a clockwise or counterclockwise manner around the element. In the example above, we chose to start on the right side of the element and then go around the element filling in dots in a clockwise direction.
Please refer to this video for a tutorial on how to "dot" your elements properly.
Moving onto drawing Lewis dot structures of various compounds:
Step 1: Count the total number of valence electrons in the compound. Use the periodic table to figure out how many valence electrons each individual element of the compound contributes then add them all up. Take H2O for example. Each hydrogen atom contributes 2 valence electrons and the oxygen atom contributes 6… for a grand total of 8 valence electrons.
Step 2: If we're dealing with a polyatomic ion with a negative charge (CO32-), add the necessary electrons to Step 1 to account for the charge. For (CO32-) we would add two more electrons or dots because each electron is equal to one unit of negative charge. Alternatively, if the polyatomic ion has a positive charge (NH4+), subtract the necessary electrons to Step 1 to account for the positive charge. For (NH4+), we would subtract one electron.
Step 3: Draw the skeletal structure of the molecule by placing a line (representing two electrons shared) between each pair of bonded atoms. Example: H—Cl.
If the molecule contains more than 2 atoms the least electronegative atom will usually be in the central position. In general, electronegativity increases from left to right on the periodic table (except for noble gases…they're perfect just the way they are). For example, carbon is less electronegative than oxygen. For the (CO32-) molecule, carbon would be the central atom. Below there are examples to see in case the visual will help.
Step 4: Next arrange the atoms in such a way where the surrounding atoms each have a complete outer shell of electrons.
Step 5: Finally add the remaining electrons to the central atom.
Step 6: Now take a look at the central atom. If the central atom has a complete octet (8 electrons), congrats, you're done. If the central atom has fewer than eight electrons, remove and electron pair from an outer atom and add another bond between that outer atom and the central atom. We have a double bond now, and our central atom now has 8 electrons.
In Olympic diving, divers hope for a perfect 10. With the outer shells of valence electrons, the perfect score is 8.
Drawing Lewis dot structures is easy, after you've practiced.
Note: Remember that hydrogen and helium can only have a maximum of 2 electrons in their outer shell.
The total number of valence electrons in this compound will be:
Carbon (4 valence e-) + four Hydrogens (4 x 1 valence e-) + Oxygen (6 valence e-) = 14 total valence electrons
Carbon is less electronegative than oxygen, so carbon will be our central atom. The individual Lewis dot structure for each atom looks like this:

If we follow our rules, we find that the only way an acceptable Lewis dot structure can be drawn is if carbon shares electrons with 3 of the hydrogen atoms and the single oxygen atom. The leftover hydrogen will have to share one pair of electrons with the oxygen.
It is important to note that electrons may easily pass from one position around an element to another, as long as they keep their pairs constant. For instance, the Lewis dot structure for oxygen can take on any of the following notations:

Putting it all together, let's arrange each of the elements and their Lewis dots around the central carbon atom:

Or we use a single line to represent an electron bond pair between the sharing atoms to look like this:

Imagine the electrons are still there on either side of the line (dashed circle), but now the line itself represents the electrons shared. Just to be sure, we can count the total number of valence electrons to make sure it adds up to the 14 we calculated above.
The final Lewis dot structure for CH4O would look like this:

Now let's try drawing the electron dot structure of a polyatomic ion, such as CO32-.
So the total number of valence electrons in this compound will be:
Carbon (4 valence e-) + three Oxygen (3 × 6 valence e-) + 2 extra e- (to account for the negative 2 charge) = 24 total valence electrons
Again, since carbon is less electronegative than oxygen, carbon will be our central atom, surrounded by the three oxygen atoms.
The lewis dot structures of each individual atom are drawn as:
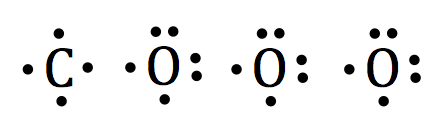
The three oxygen atoms will be surrounding the carbon atom.

Now let's draw the remaining valence electrons around the oxygen atoms.

Now that all 24 electrons have been drawn on the Lewis dot structure, look at the central carbon atom. Notice how it doesn't have a full octet. As drawn it only has 6 valence electrons, so it needs to form another bond with one of the oxygen atoms in order to complete its octet. We will take an electron pair away from one of the oxygen atoms and make a double bond with carbon instead.

Since it is a polyatomic ion, we place a bracket around the entire structure and add the charge of negative two to the outside for the complete and final Lewis Dot Notation of CO32-.

Note that the double bond may be placed on any one of the oxygen atoms. In fact, when a double bond can shift from one atom to another, like in the case above, the molecular structure can be represented by more than one Lewis structure. This phenomenon is referred to as resonance and adds increased stability to the molecule. In the case above, there are three potential resonance forms that are considered to exist simultaneously.
All this talk about sharing electrons between elements make us want to change your name to Bond.
The orbital notation also helps us figure out what's really going on inside all of those crazy orbitals. For instance, when we see the electron configuration "3p5," we know that there are 5 electrons in the p subshell, but we don't know which orbitals the electrons are actually located in. Not to worry, the orbital notation provides us with that level of detail that simply can't be found in the electron configuration.
Let's get started. First we need to do the electron configuration of an atom. Want to use silicon as an example? We hope so.
Sample Problem
Silicon1s22s22p63s23p2
First let's recall:
• s subshells have 1 orbital
• p subshells have 3 orbitals
• d subshells have 5 orbitals
• f subshells have 7 orbitals
For each subshell we'll first draw the corresponding number of orbitals (circles) above them as shown here:

Next we'll fill in the electrons represented as arrows. Don't forget Hund's Rule when filling in your electrons. Remember that the most stable arrangement for electrons in each subshell is the one with the greatest number of parallel spins, so we will distribute electrons evenly to orbitals with a given subshell first before any pairing occurs. In other words, each circle gets one arrow before any circle gets two arrows.

There are two other ways that we might see the orbital notation represented:
1) Where the orbitals are represented as rectangles and the electrons are represented as arrows.

2) Where the orbitals are represented as circles and the electrons are represented as slashes.

Lewis Dot Notation
There's another special and common notation in chemistry that we will master: Lewis Dot Notation.Now presenting, direct from Berkley, California: Lewis and his Amazing Dots. Lewis dot notation was first presented by Gilbert Newton Lewis in 1916 in an article entitled "The Atom and the Molecule" published in the Journal of the American Chemical Society.

This is going to look simple. That's because it is simple; thanks to good ol' Gilbert. Lewis was the first person we know to have done this and make it so obviously simple for the rest of us. Good job, Lewis.
This notation is just another way of showing details about an atom's valence (outer) shell. Unlike the electron configuration, which includes all of the electrons an atom has to offer, Lewis dots are only concerned with electrons in the outer most shell of an atom. This means that no matter how many electrons an element contains, Lewis dots will never number higher than eight. If it helps, just remember that super hip TV show from 1977 Eight is Enough.
Let's go back to silicon as an example. We've already learned the electron configuration of silicon is 1s22s22p63s23p2. The first (n = 1) and second (n = 2) shells are completely filled. The third (n = 3) shell has a filled s orbital but it's p and d orbitals remain empty. Therefore the third shell is our valence shell and the electrons in this shell are the valence electrons.
The Lewis dot notation consists of the kernel (the nucleus and all of its inner electrons that is represented by the elemental symbol) and the valence electrons represented by dots. Check out the lewis dot structure of carbon below:

The letter "C" is the kernel, and as we said this represents the atom and its inner shell electrons (the 1s2 electrons). The remaining four valence electrons are placed around the kernel according to specific rules. Don't worry about the rules just yet. We'll get to them soon. Each of the four sides around the elemental symbol represents one of the orbitals in the outer shell. Here are the rules:
Rule #1. No side can have more than two dots because each orbital can only hold two electrons.
Rule #2. When filling the sides of the element symbol each side gets one dot before doubling up…that's Hund's Rule, not ours. The exceptions to this rule are hydrogen and helium since their outer shells can only hold two electrons. Every other element obeys the second rule.
How many dots should we place around each element? The answer is surprisingly simple. Just take a look at the periodic table below. Remember how each dot is supposed to represent the number of electrons in the elements outer shell? Well, the periodic table displays the elements in a way for us to easily determine how many electrons are in their outer shell.
The column in which the element is located will tell us how many valence electrons each element has in its outer shell. Remember hydrogen and helium can only have a maximum of 2 valence electrons in their outer shell so they are the exceptions to the rule.

Let's see some examples:

As we go straight across the second row of the periodic table we keep adding one more dot or electron (clockwise) to each element. We can do this all the way to Ne, the noble gas, which has a full octet and is quite pleased about it, too.
Keep in mind it doesn't matter where we first put our dot around the element. Just make sure you choose a side and then start adding your dots in either a clockwise or counterclockwise manner around the element. In the example above, we chose to start on the right side of the element and then go around the element filling in dots in a clockwise direction.
Please refer to this video for a tutorial on how to "dot" your elements properly.
Moving onto drawing Lewis dot structures of various compounds:
Step 1: Count the total number of valence electrons in the compound. Use the periodic table to figure out how many valence electrons each individual element of the compound contributes then add them all up. Take H2O for example. Each hydrogen atom contributes 2 valence electrons and the oxygen atom contributes 6… for a grand total of 8 valence electrons.
Step 2: If we're dealing with a polyatomic ion with a negative charge (CO32-), add the necessary electrons to Step 1 to account for the charge. For (CO32-) we would add two more electrons or dots because each electron is equal to one unit of negative charge. Alternatively, if the polyatomic ion has a positive charge (NH4+), subtract the necessary electrons to Step 1 to account for the positive charge. For (NH4+), we would subtract one electron.
Step 3: Draw the skeletal structure of the molecule by placing a line (representing two electrons shared) between each pair of bonded atoms. Example: H—Cl.
If the molecule contains more than 2 atoms the least electronegative atom will usually be in the central position. In general, electronegativity increases from left to right on the periodic table (except for noble gases…they're perfect just the way they are). For example, carbon is less electronegative than oxygen. For the (CO32-) molecule, carbon would be the central atom. Below there are examples to see in case the visual will help.
Step 4: Next arrange the atoms in such a way where the surrounding atoms each have a complete outer shell of electrons.
Step 5: Finally add the remaining electrons to the central atom.
Step 6: Now take a look at the central atom. If the central atom has a complete octet (8 electrons), congrats, you're done. If the central atom has fewer than eight electrons, remove and electron pair from an outer atom and add another bond between that outer atom and the central atom. We have a double bond now, and our central atom now has 8 electrons.
In Olympic diving, divers hope for a perfect 10. With the outer shells of valence electrons, the perfect score is 8.
Drawing Lewis dot structures is easy, after you've practiced.
Sample Problem
Draw the Lewis structural formula for CH4O.Note: Remember that hydrogen and helium can only have a maximum of 2 electrons in their outer shell.
The total number of valence electrons in this compound will be:
Carbon (4 valence e-) + four Hydrogens (4 x 1 valence e-) + Oxygen (6 valence e-) = 14 total valence electrons
Carbon is less electronegative than oxygen, so carbon will be our central atom. The individual Lewis dot structure for each atom looks like this:

If we follow our rules, we find that the only way an acceptable Lewis dot structure can be drawn is if carbon shares electrons with 3 of the hydrogen atoms and the single oxygen atom. The leftover hydrogen will have to share one pair of electrons with the oxygen.
It is important to note that electrons may easily pass from one position around an element to another, as long as they keep their pairs constant. For instance, the Lewis dot structure for oxygen can take on any of the following notations:

Putting it all together, let's arrange each of the elements and their Lewis dots around the central carbon atom:

Or we use a single line to represent an electron bond pair between the sharing atoms to look like this:

Imagine the electrons are still there on either side of the line (dashed circle), but now the line itself represents the electrons shared. Just to be sure, we can count the total number of valence electrons to make sure it adds up to the 14 we calculated above.
The final Lewis dot structure for CH4O would look like this:

Now let's try drawing the electron dot structure of a polyatomic ion, such as CO32-.
So the total number of valence electrons in this compound will be:
Carbon (4 valence e-) + three Oxygen (3 × 6 valence e-) + 2 extra e- (to account for the negative 2 charge) = 24 total valence electrons
Again, since carbon is less electronegative than oxygen, carbon will be our central atom, surrounded by the three oxygen atoms.
The lewis dot structures of each individual atom are drawn as:
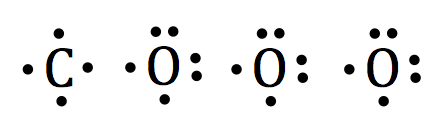
The three oxygen atoms will be surrounding the carbon atom.

Now let's draw the remaining valence electrons around the oxygen atoms.

Now that all 24 electrons have been drawn on the Lewis dot structure, look at the central carbon atom. Notice how it doesn't have a full octet. As drawn it only has 6 valence electrons, so it needs to form another bond with one of the oxygen atoms in order to complete its octet. We will take an electron pair away from one of the oxygen atoms and make a double bond with carbon instead.

Since it is a polyatomic ion, we place a bracket around the entire structure and add the charge of negative two to the outside for the complete and final Lewis Dot Notation of CO32-.

Note that the double bond may be placed on any one of the oxygen atoms. In fact, when a double bond can shift from one atom to another, like in the case above, the molecular structure can be represented by more than one Lewis structure. This phenomenon is referred to as resonance and adds increased stability to the molecule. In the case above, there are three potential resonance forms that are considered to exist simultaneously.
All this talk about sharing electrons between elements make us want to change your name to Bond.