The Periodic Table
The periodic table is a chemist's best friend. It will also be our BFF for much of this course. Tried and true, it will illuminate the path to chemical enlightenment, or at the very least help with understanding general chemistry.
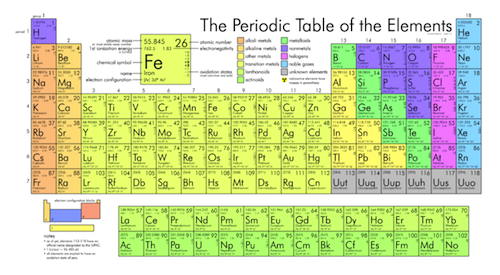
The periodic table, or Rosetta stone of chemistry, is shown. See larger image here.
All known elements can be found on the periodic table. Each element has its own special symbol. For example, the symbol for carbon is "C." The symbol for oxygen is "O." The symbol for hydrogen is "H." It seems simple enough until we come across an element such as sodium. The chemical symbol for sodium is "Na." Nah, we aren't kidding. Some symbols are assigned based on the element's Latin name. In the case of sodium, its Latin name is Natasha. Just joking. Natrium is sodium's Latin name. Not even we could make that one up.
Check out these cool interactive periodic tables for a more in-depth look at all of the elements and trivia here and here.
Each element on the periodic table is also assigned a number called the atomic number. The atomic number is the number of protons in every atom of that particular element. Remember we said that we would come back to this? An atom is identified as a particular element based on the number of protons it has.
Take a look at a periodic table. Notice that the atomic numbers increase by one as we go from left to right. This is not by chance. The periodic table is organized such that elements are arranged in order of increasing numbers of protons.
In fact, there are all sorts of what scientists call "trends" in the organization of the periodic table. These are not fashion trends like boot cut or skinny jeans. The elements are always in fashion when it comes to chemistry. (Corny, we know.) These trends have more to do with how elemental matter behaves. Let's look at an example.
Vertical columns of the periodic table comprise what is called a family. This is not your typical family with a parental unit, some rug rats, and a pet. This is a chemical family. Chemical families share chemical properties because of similarities in atomic composition.
One of our favorite families is the family in the first vertical column of the periodic table. This family is called the alkali metals family. The reason why we like them is that they share a tendency for explosiveness.
Check out what happens when you add members of this family to water. It turns out that the family members with the largest atomic numbers are the most reactive with water.
The periodic table is unlike any other chart or table we've seen. It can be read in multiple directions and holds a wealth of knowledge. It helps us predict trends and is a fantabulous resource. The key to chemistry it is.

The periodic table, or Rosetta stone of chemistry, is shown. See larger image here.
All known elements can be found on the periodic table. Each element has its own special symbol. For example, the symbol for carbon is "C." The symbol for oxygen is "O." The symbol for hydrogen is "H." It seems simple enough until we come across an element such as sodium. The chemical symbol for sodium is "Na." Nah, we aren't kidding. Some symbols are assigned based on the element's Latin name. In the case of sodium, its Latin name is Natasha. Just joking. Natrium is sodium's Latin name. Not even we could make that one up.
Check out these cool interactive periodic tables for a more in-depth look at all of the elements and trivia here and here.
Each element on the periodic table is also assigned a number called the atomic number. The atomic number is the number of protons in every atom of that particular element. Remember we said that we would come back to this? An atom is identified as a particular element based on the number of protons it has.
Take a look at a periodic table. Notice that the atomic numbers increase by one as we go from left to right. This is not by chance. The periodic table is organized such that elements are arranged in order of increasing numbers of protons.
In fact, there are all sorts of what scientists call "trends" in the organization of the periodic table. These are not fashion trends like boot cut or skinny jeans. The elements are always in fashion when it comes to chemistry. (Corny, we know.) These trends have more to do with how elemental matter behaves. Let's look at an example.
Vertical columns of the periodic table comprise what is called a family. This is not your typical family with a parental unit, some rug rats, and a pet. This is a chemical family. Chemical families share chemical properties because of similarities in atomic composition.
One of our favorite families is the family in the first vertical column of the periodic table. This family is called the alkali metals family. The reason why we like them is that they share a tendency for explosiveness.
Check out what happens when you add members of this family to water. It turns out that the family members with the largest atomic numbers are the most reactive with water.
The periodic table is unlike any other chart or table we've seen. It can be read in multiple directions and holds a wealth of knowledge. It helps us predict trends and is a fantabulous resource. The key to chemistry it is.