The Carbon Cycle
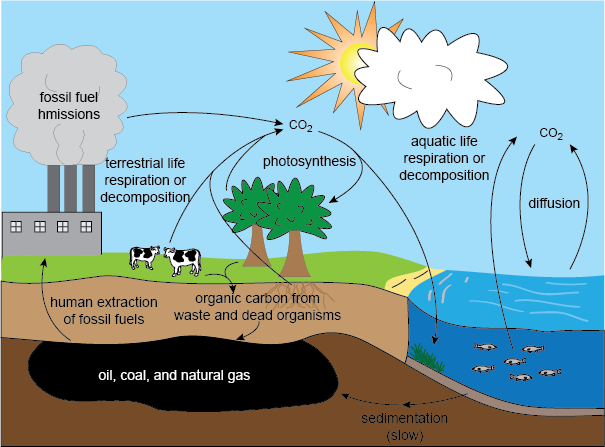
Carbon is likely the next most important chemical in an ecosystem. Most of the carbon on Earth is found in the atmosphere in the form of carbon dioxide, or CO2.
Plants and other producers extract this gaseous carbon and, using energy from the sun, convert it into sugar. Producers and consumers alike use this sugar form of carbon as a source of energy and as a source of raw material for growth. As carbon moves through the food chain, much of it is released back into the atmosphere as CO2 when consumers exhale. Whatever is not exhaled eventually accumulates in the ground when organisms die and decompose. Once carbon is in the ground, it leaves the carbon cycle forever. The exception to this is when humans extract it in the form of oil, coal, and natural gas, and release it back into the atmosphere by burning it. The endpoint here is that without carbon, producers would not have any way to use energy from the sun. In fact, life as we know it wouldn’t exist at all. And that would suck just a little bit.
Brain Snack
You've probably heard that CO2 is a serious greenhouse gas, but methane, CH4, is 20 times as potent. Methane is produced naturally by a group of microbes called methanogens. These microbes live deep in anoxic sediments, associated with plants, and also, yes, in cows. The average cow releases somewhere between 500-600 liters of methane into the air per day! Holy cow!
Plants and other producers extract this gaseous carbon and, using energy from the sun, convert it into sugar. Producers and consumers alike use this sugar form of carbon as a source of energy and as a source of raw material for growth. As carbon moves through the food chain, much of it is released back into the atmosphere as CO2 when consumers exhale. Whatever is not exhaled eventually accumulates in the ground when organisms die and decompose. Once carbon is in the ground, it leaves the carbon cycle forever. The exception to this is when humans extract it in the form of oil, coal, and natural gas, and release it back into the atmosphere by burning it. The endpoint here is that without carbon, producers would not have any way to use energy from the sun. In fact, life as we know it wouldn’t exist at all. And that would suck just a little bit.

Brain Snack
You've probably heard that CO2 is a serious greenhouse gas, but methane, CH4, is 20 times as potent. Methane is produced naturally by a group of microbes called methanogens. These microbes live deep in anoxic sediments, associated with plants, and also, yes, in cows. The average cow releases somewhere between 500-600 liters of methane into the air per day! Holy cow!